Negative pressure wound therapy (NPWT) has been used traditionally in acute care settings for more than 15 years, and more recently, within primary care. However, the need to make appropriate funding arrangements, has often led to a disruption in therapy during transfer of the patient from the acute setting to primary care. The availability on Drug Tariff of the V.A.C.Via Negative Pressure Wound Therapy System (KCI) a new off-the-shelf, disposable NPWT device has revolutionised financial planning across acute and primary services, minimising delay at discharge. This can help to mobilise patients earlier and shorten the length of their hospital stay, reducing overall treatment costs. Additionally, V.A.C.Via Therapy can be commenced in the community and may negate the need for hospital admission.
What is V.A.C.® Therapy?
Negative pressure wound therapy (NPWT) also known as topical negative pressure (TNP) or vacuum-assisted closure (V.A.C.® Therapy, KCI) has been used in wound care since 1997 (Argenta and Morykwas, 1997; EWMA, 2007; WUWHS, 2008), when the first device was cleared by the US Food and Drug Administration (FDA) for marketing in the USA.
V.A.C.® Therapy is the most established negative pressure system and uses a vacuum to create continuous or intermittent negative pressure at the wound bed to remove fluid, exudate and infectious materials as well as stimulating perfusion and new tissue growth (WUWHS, 2008) (Box 1).
What is V.A.C.Via™ therapy?
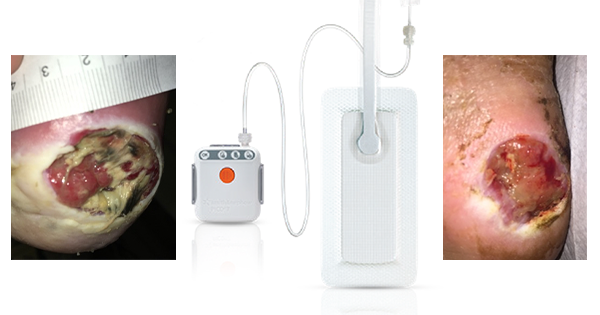
The V.A.C.Via™ Therapy unit is a new single-patient use, disposable NPWT system that uses a one-button operating system. The simple interface allows the user to select the pressure setting and therapy mode, and to turn the therapy on.
The device is portable, lightweight (it weighs 0.32kg when the canister is empty) and is small in size, allowing it to be worn under clothing, be put into a pocket of a jacket or attached to a belt at the waist using the integral clip. The therapy unit has a 7-day lifespan and includes a rechargeable battery. Replacement therapy units can be purchased to continue therapy for longer than seven days.
Benefits of V.A.C.Via™ Therapy include:
- Offers V.A.C® Therapy in a smaller therapy unit
- Allows improved continuity of care between care settings
- Patients can receive seven days of therapy at home or in hospital
- It is safe and easy to use
- It is lightweight and portable
- The device is virtually silent and can be worn discretely.
Key features of V.A.C.ViaTM therapy
The V.A.C.Via™ Therapy System incorporates a number of established technologies used for standard V.A.C.® Therapy devices. The unit is compatible with a new Spiral GranuFoamTM Dressing, which has the same hydrophobic structure of conventional GranuFoamTM. This helps to promote granulation in the wound bed through microdeformation of cells. It can be cut and adapted to fit the contours of the wound before applying the drape to seal the wound.
V.A.C.Via™ Therapy also incorporates SensaT.R.A.C.™ Technology. This intelligent system continually measures pressures at the wound site and automatically compensates for pressure variances due to changes in the patient’s position, movement and level of exudate (McNulty et al, 2010). This maintains the prescribed level of pressure at the wound site.
In patients with diabetic foot wounds, SensaT.R.A.C.™ Technology is important in preventing a pressure drop where there is a vertical height difference between the location of the dressing (foot) and the position of the therapy unit (waist). The main unit also provides safety alarms that alert clinicians if the target pressure is not maintained or there is a blockage in the tubing.
In addition, V.A.C.Via™ Therapy incorporates several innovative technologies that have been specially developed for this device.
Dynamic Pressure Control™ Therapy
V.A.C.Via™ Therapy delivers continuous sub-atmospheric pressure (-75mmHg or -125mmHg) or Dynamic Pressure Control™ (DPC) Therapy. DPCTM Therapy offers intermittent therapy in six minute cycles that rotates between the target pressure (either -75mmHg or-125mmHg) and a minimum low level of negative pressure (-25mmHg). The action of keeping the pressure above 0mmHg between negative pressure cycles helps reduce any discomfort associated with foam dressing expansion and helps to prevent dressing leakage, which can occur in the absence of negative pressure. The continuous mode should be used for the first 48 hours in all wounds, as recommended in the therapy guidelines.
Vortis™ Pump Technology
This acoustic resonance pump harnesses sound waves at a frequency inaudible to most humans, allowing therapy to be applied almost silently. The pump is smaller than a £2 coin and each unit contains two pumps.
When is V.A.C.Via™ therapy indicated?
The V.A.C.Via™ Therapy System is designed for low to moderately exuding wounds, including:
Sub-acute and dehisced wounds
Venous leg, diabetic foot and pressure ulcers
Flaps and grafts
Small to moderate acute wounds prior to definitive closure.
Although V.A.C.Via™ Therapy is ideal for use in low to moderately exuding wounds, it incorporates a 250ml canister, which means it can be used on small wounds with high levels of exudate, e.g. over a skin graft in patients with lymphoedema following excision of a lesion.
V.A.C.Via™ Therapy can also be used as a temporising dressing to maintain a sealed, clean environment prior to definitive closure in lower limb trauma. This allows the patient to be discharged home and readmitted for surgery at a later date.
For large wounds, treatment with a standard NPWT device should be considered to reduce the wound size and reapproximate the wound edges prior to the use of V.A.C.Via™ Therapy.
Key considerations in choosing V.A.CVia™ Therapy over standard NPWT include:
- Wounds that are small and less complex
- Patient can be mobilised early
- Suitability for discharge home
- To prevent admission to hospital.
As an off-the-shelf product, V.A.CVia™ Therapy can also allow early initiation of NPWT, which may be associated with a shorter length of hospital stay (Baharestani et al, 2008).
How to apply V.A.C.Via™ Therapy
Prior to application:
A full assessment should be undertaken to establish the patient’s age, level of mobility and independent living, as well as address any underlying medical conditions, including glucose control, fluid balance and the provision of adequate analgesia.
All wounds should be debrided to remove non-viable tissue before use and any infection managed according to local protocols (Henderson et al, 2010).
Patients should be evaluated for bleeding risk (FDA, 2010), but it is also important that the wound has an adequate blood supply.
Step by step guide to application:
- Prepare the wound and apply a barrier film dressing if required to protect the periwound skin.
- Apply the Spiral GranuFoam™ Dressing. Trim foam away from the wound and coil gently (do not pack) into the wound. Do not overlay onto the peripheral wound area as this will hinder wound edge approximation and ensure the dressing sits within the wound margins.
- Trim the drape to cover the dressing with a border of at least 3-5cm at the wound edge to allow contact with intact periwound skin. Cut a 2.5cm hole in the drape and place the SensaT.R.A.C.™ Pad on top. Apply light finger pressure to create a good seal.
- Engage canister on the back of the therapy unit and press the power button to commence therapy.
How frequently should the dressings be changed?
For wounds that are not infected, the dressing should be changed once every 48-72 hours or at least three times per week as the wound reduces in size. Infected wounds may require more frequent dressing changes.
Patients, who are treated at home should not change or remove their own dressings and/or interfere with the therapy settings.
Using the device at home
During the day the therapy unit can be worn beneath clothing. It is recommended that it is plugged in during sleep and placed so that the tubing will not become kinked. The patient may wash or shower with the dressing in place, taking care not to soak the dressing. The tube should be clamped and disconnected from the therapy unit. Although simple to use, patients and carers should have training on how to operate the device safely and recognise any signs and symptoms that require immediate clinical attention.
Patients should be supplied with the instructions for using the device together with the name of the local contact person. A 24-hour helpline, managed in the UK, is available to support patients and carers.
When to stop V.A.C.Via™ therapy?
Treatment can be stopped when the objective of the treatment has been achieved. The goal of therapy can vary from preparation for surgical closure, healing by secondary intention or bolstering of a skin flap.
What is the evidence for use?
The evidence base for V.A.C.® Therapy is well established both in acute and chronic wounds in adults (WUWHS, 2008), with more than 25 randomised controlled trials published to date.
Acute wounds
The efficacy of early, short-term NPWT in preparing acute wounds for surgical closure has been extensively researched (DeFranzo et al, 2001; Rinker et al, 2008; Kaplan et al, 2009; Stannard et al, 2010). Two studies comparing the effect of V.A.C.® Therapy to moist wound care after partial foot amputation found that more patients healed with V.A.C.® Therapy and/or at a faster closure rate (Armstrong and Lavery, 2005; Armstrong et al, 2007). More recently, Paola et al (2010) ran two study groups: one investigating the effectiveness of V.A.C.® Therapy in improving graft takes on diabetic foot wounds and the other assessing its effect on infected open minor amputations. The findings of both studies demonstrated that V.A.C.® Therapy results in faster wound bed preparation and closure and improved rates of graft take, compared to standard treatment.
Chronic wounds
Joseph et al (2000) compared V.A.C.® Therapy with moist wound therapy in 24 patients with chronic, non-healing wounds. This reported a significant reduction in the volume of the wounds treated with V.A.C.® Therapy (66% vs 20%; p<0.0001). In a later study by Vuerstaek et al (2006), the time to healing in 60 leg ulcer patients revealed a shorter healing time for V.A.C.® Therapy when compared with conventional therapy ( 29 vs 45 days). Blume at al (2008) investigated the efficacy of V.A.C.® Therapy versus advanced moist wound therapy in 342 diabetic foot ulcer patients. A higher percentage of patients achieved wound closure with V.A.C.® Therapy within the 112-day treatment period and fewer amputations were reported.
Cost/benefits of V.A.C.® Therapy
Apelqvist et al (2008) found that the use of NPWT in post-amputation wounds was associated with less resource use and lower overall cost of care compared to moist wound healing. Flack et al (2008) also found that V.A.C.® Therapy was more effective than both traditional and advanced dressings in patients with diabetic foot ulcers, resulting in reduced costs. Patient comfort was also found to be an advantage in those receiving V.A.C.® Therapy (Braakenburg et al, 2006).
How can I obtain V.A.C.Via™ therapy?
V.A.C.ViaTM Therapy is a purchase model and is not available as a rental device. It is currently available in the UK on Drug Tariff, through wholesalers and direct from the manufacturer. It is supplied as a Starter Kit (containing the therapy unit, one Spiral GranuFoam™ Dressing kit, canister, charger and carrying case) or 7-day kit (containing the therapy unit, three Spiral GranuFoam™ Dressing kits, two canisters, charger and carry case). Additional dressing kits and canisters are also available.
Summary
V.A.C.Via™ Therapy is a next generation NPWT device that incorporates the proven actions of V.A.C.® Therapy in a small, user-friendly unit. It has been developed using the virtually silent Vortis™ Pump Technology and provides continuous or DPC™ negative pressure at the wound bed to promote the formulation of granulation tissue and wound healing. It can be initiated quickly and is ideal for patients who need to continue therapy at home, which can help to reduce time spent in hospital, promote earlier mobilisation and allow patients to return to normal daily activities faster.
AUTHOR DETAILS
Stephen-Haynes J[1], Woodrow T[2], Wharton S[3]
- Visiting Professor in Tissue Viability, Professional Development Unit, Birmingham City University and Consultant Nurse, Worcestershire Health & Care Trust
- Specialist Podiatrist Diabetes, Cornwall and Isles of Scilly Community Health Services
- Consultant Plastic and Reconstructive Surgeon, Russells Hall Hospital, Dudley
References
- Argenta LC, Morykwas MJ (1997) Vacuum-assisted closure: a new method for wound control and treatment: clinical experience. Ann Plast Surg 38(6): 563-76
- Apelqvist J, Armstrong DG, Lavery L, Bouton AJM (2008) Resource utilization and economic cost of care based on a randomised trial of vacuum-assisted closure therapy in the treatment of diabetic foot wounds. Am J Surgery 195;782-8
- Armstrong DG, Lavery LA; Diabetic Foot Study Consortium (2005) Negative pressure wound therapy after partial diabetic foot amputation: a multicentre, randomised controlled trial. Lancet 366(9498):1704-10
- Armstrong DG, Marston WA, Boulton AJ (2007) Negative pressure wound therapy via vacuum-assisted closure following partial foot amputation: what is the role of wound chronicity? Int Wound J 4(1):79-86
- Baharestani MM, Houliston-Otto DB, Barnes S (2008) Early versus late initiation of negative pressure would therapy: examining the impact on home care length of stay. Ostomy Wound Manage 54(11):48-53
- Blume PA, Walters J, Payne W, et al (2008) Comparison of negative pressure wound therapy using vacuum-assisted closure with advanced moist wound therapy in the treatment of diabetic foot ulcers: a multicenter randomized controlled trial. Diabetes Care 31(4):631-6
- Braakenburg A, Obdeijn MC, Feitz R, et al (2006) The clinical efficacy and cost effectiveness of the vacuum-assisted closure technique in the management of acute and chronic wounds: a randomized controlled trail. Plast Reconstr Surg 118(2): 390-7
- DeFranzo AJ, Argenta LC, Marks MW, et al (2001) The use of vacuum-assisted closure therapy for the treatment of lower-extremity wounds with exposed bone. Plast Reconstr Surg 108(5): 1184-91
- EWMA (2007) Position Document Topical negative pressure in wound management. MEP Ltd, London
- FDA (2010) Negative pressure wound therapy: use with care. Available at: http://www.fda.gov/MedicalDevices/Safety/AlertsandNotices/TipsandArticlesonDeviceSafety/ucm225038.htm [accessed October 2011].
- Flack S, Apelqvist J, Keith M et al (2008) An economic evaluation of VAC therapy compared with wound dressings in the treatment of diabetic foot ulcers. J Wound Care 17(2): 71-8.
- Henderson V, Timmons J, Hurd T, et al (2010) NPWT in everyday practice made easy. Wounds International 2010; 1(5): Available from http://www.woundsinternational.com
- Joseph E, Hamori C, Bergman S, et al (2000) A new prospective randomized trial of vacuum assisted closure versus standard therapy of chronic non-healing wounds. Wounds 12:60-7
- Kaplan M, Daly D, Stemkowski S (2009) Early intervention of negative pressure wound therapy using Vacuum-Assisted Closure in trauma patients: impact on hospital length of stay and cost. Adv Skin Wound Care 22(3):128-32
- McNulty A, Spranger I, Courage J, et al (2010) The consistent delivery of negative pressure to wounds using reticulated, open cell foam and regulated pressure feedback. Wounds 22(5):114-20
- Paola LD, Carone A, Ricci S, et al (2010) Use of vacuum assisted closure therapy in the treatment of diabetic foot wounds. J Diabet Foot Complications 20(2):33-44
- Pfau M, Rennekampff HO, Schaller HE (2000) Skin graft fixation by vacuum assisted topical foam dressing. J Burn Care Rehab 21(1): 1
- Rinker B, Amspacher JC, Wilson PC, Vasconez HC (2008) Subatmospheric pressure dressing as a bridge to free tissue transfer in the treatment of open tibia fractures. Plast Reconstr Surg 121(5):1664-73
- Stannard JP, Singanamala N, Volgas DA (2010) Fix and flap in the era of vacuum suction devices: what do we know in terms of evidence based medicine? Injury 41(8):780-6
- Schneider AM, Morykwas MJ, Argenta LC (1998) A new and reliable method of securing skin grafts to the difficult recipient bed. Plast Reconstr Surg 102(4):1195-8
- WUWHS (2008) Principles of best practice: Vacuum assisted closure: recommendations for use. A consensus document. MEP Ltd, London
- Vuerstaek JD, Vainas T, Wuite J, et al (2006) State-of-the-art treatment of chronic leg ulcers: A randomized controlled trial comparing vacuum-assisted closure (V.A.C.) with modern wound dressings. J Vasc Surg 44(5):1029-37; discussion 1038