Projections from the Health Foundation (2023) indicate the prevalence of major health conditions will rise from fewer than one in six individuals in 2019 to approximately one in five by 2040, with increasing numbers of children and working-age adults experiencing long-term health conditions. The Office for Budget Responsibility (2023) forecasts health expenditure will escalate from approximately 8% of GDP in 2024–2025 to 14.5% by 2073.
The NHS 10-year plan, published in July 2025 (UK Government, 2025), highlighted three major changes:
- Moving from hospital to community care, with neighbourhood health centres available in every community.
- A shift from analogue to digital using new technology, reducing administration.
- A move away from a sickness model to one of ill health prevention.
To fulfil their strategic responsibilities effectively, Integrated Care Boards are expected to develop a range of capabilities tailored to system-wide healthcare transformation:
- Excellent analytical capability, to be guided by population health data;
- A strong strategy function, including staff with good problem-solving and analytical skills;
- Capability in partnership working and an understanding of value-based healthcare;
- Intelligent healthcare payer understanding, to support a focus on value for money, the development of novel payment mechanisms and oversight of strategic resource allocation;
- User involvement functions to ensure services meet the needs of communities.
The 10-year plan states: the range of clinical tasks done by nurses has increased to include prescribing, some diagnosis and assessment, management of complex wounds and administration of intravenous therapies. The plan commits to advanced practice models for nurses, midwives and allied health professionals, with wound care cited as an area requiring accreditation and regulation. The emphasis on multidisciplinary neighbourhood health centres presents the possibility for integration of specialist wound care services.
With an increasing ageing population and an increase in long-term conditions (including complex wounds), evidence-based practice (EBP) must be implemented in wound care practice to improve outcomes, reduce cost, reduce unnecessary healthcare visits, reduce needless waste and improve sustainability.
Evidence-based practice
EBP enhances the quality of healthcare, reduces costs, improves patient outcomes, empowers clinicians and is recognised as a problem-solving approach (Mazurek Melnyk and Fineout-Overholt, 2019). It integrates the best available clinical evidence with professional expertise and patient values and preferences. By combining clinical judgement, current research and the patient’s individual needs, EBP supports consistent decision-making and optimised care delivery (Abu-Baker et al, 2021). Findings from a scoping review concluded that implementation of EBP results in improved patient outcomes and healthcare return on investment (Connor et al, 2023). Additionally, Solberg et al (2018) emphasised that EBP provides clinical researchers with the most robust available evidence to analyse and make informed decisions in supporting clinical campaigns, programmes or policies to improve the health and well-being of populations. Through the integration of robust clinical evidence, practitioner expertise and individual patient values, EBP establishes a fundamental framework for the standardisation of clinical care (Abu-Baker et al, 2021).
EBP-informed strategies in wound care, such as biofilm-based wound care, antimicrobial stewardship and targeted debridement, have demonstrated improvements in healing rates, reductions in infection and enhanced resource efficiency (Kilroy-Findley and Ousey, 2023; Dowsett et al, 2021). Nevertheless, considerable variation persists in wound assessment and treatment, often attributable to the underuse of evidence-based interventions and suboptimal clinical decision-making (Gray et al, 2019; Murray and Norrie, 2020), which can compromise the healing trajectory for patients, contributing to the development of hard-to-heal wounds.
Implications of unwarranted variation in wound care
Hard-to-heal wounds place a considerable financial strain on healthcare systems through increasing demands on clinical staffing, dressing use and pharmacological interventions. The burden on patients is equally significant, encompassing loss of income, increased personal healthcare costs, exacerbated pain, psychological and physical distress, reduced mobility, sleep disturbances and social isolation (Beeckman et al, 2023).
Unwarranted variation in wound care practices directly impacts patient outcomes, contributing to increased healthcare costs and prolonged healing durations (National Wound Care Strategy Programme, 2023). Addressing this inconsistency presents significant opportunities to improve healing trajectories, reduce recurrence rates, alleviate patient suffering, and reduce spending on inappropriate or ineffective interventions. Variability in the quality and availability of clinical evidence necessitates flexibility in wound care practice, with clinicians often required to make informed decisions based on the best evidence currently accessible. Despite the availability of high-quality evidence, optimal or gold-standard practices are frequently underused or inconsistently implemented in clinical settings (Wounds UK, 2023). These disparities highlight the urgent need to adopt uniform, evidence-driven practices to promote high-quality, equitable wound care.
Evidence indicates that healing rates in venous leg ulcer management differ depending on how care is aligned with the evidence base, with community-based healing rates reported as low as 37% at 52 weeks (Guest, 2020) compared to rates exceeding 85% at 24 weeks in settings where care is standardised and evidence-informed (Gohel et al, 2018). Given the variability in the strength of available evidence, clinicians must remain flexible in applying existing resources while maintaining accountability when employing interventions lacking robust support.
In the absence of high-level data, potentially beneficial products may be overlooked or underused. Conversely, the application of interventions lacking robust evidence demands clear clinical accountability. Despite the existence of authoritative guidance, such as NICE recommendations and data from randomised controlled trials, the consistent delivery of evidence-based, gold-standard care remains limited in practice (Wounds UK, 2023). Equally concerning is the omission of products or approaches that are supported by well-established evidence.
Designing an evidence-based pathway to optimise clinical outcomes
The pathway was developed using the UrgoStart Plus Treatment Range [Figure 1] to enable standardised decision-making, reduce unwarranted variation and optimise resource use. The UrgoStart Plus Treatment Range was chosen due to clinical evidence supporting positive outcomes. The EXPLORER trial (Edmonds et al, 2018) demonstrated significantly higher wound closure rates in patients with diabetic foot ulcers compared to standard care (p=0.002), with the CHALLENGE trial (Meaume et al, 2012) showing improved wound area reduction and pain scores in patients with venous leg ulcers over 8 weeks. NICE (2023) concluded that, drawing on published evidence, economic modelling, and expert consensus, UrgoStart is likely to offer cost savings compared to non-interactive dressings. In the case of patients with diabetic foot ulcers, the committee agreed on a projected £342 annual saving per patient when UrgoStart is used. For patients with venous leg ulcers, the committee similarly acknowledged the potential for cost savings. However, it noted greater uncertainty around the estimates due to limitations in the evidence base for complete wound healing.
Although evidence-based, there was some resistance to the change and implementation of the pathway. Resistance to change among nurses is a well-documented phenomenon (Cheraghi et al, 2023; Clark, 2013), often arising during the introduction of new clinical practices or technologies. It is recognised that nurses may inherently resist change due to a variety of emotional and cognitive responses, including fear, uncertainty, doubt, frustration, distrust, confusion, and anger (Udod and Wagner, 2018). These reactions, though challenging, are largely predictable and reflect the complex interplay between professional identity, workload pressures, and patient care responsibilities (Clark, 2013; Cheraghi et al, 2023). To overcome this resistance, the pathway was systematically introduced across the Trust through a coordinated programme of engagement. This included targeted information sessions, structured presentations at tissue viability and link nurse meetings, and ongoing support from the Tissue Viability Team. These activities facilitated shared understanding, built confidence in the evidence base, and encouraged ownership among frontline staff.
The following case study demonstrates the effectiveness of implementing a standardised pathway.
Case Study 1: Management of a chronic leg ulcer using UrgoStart Plus and UrgoKTwo
A 67-year-old female presented with a chronic, non-healing wound with a duration of 6 months. The wound developed following an acute traumatic injury sustained while gardening. She had a medical history of chronic venous disease, obesity and hypertension.
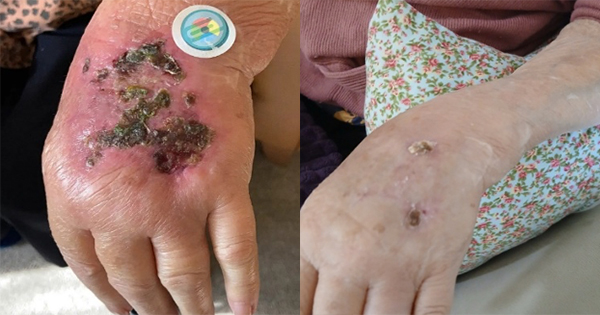
On assessment, the wound demonstrated classic signs of chronic ulceration. The affected limb showed marked oedema, limb shape distortion and haemosiderin staining [Figure 2]. Despite receiving multiple treatment regimens, including Aquacel Ag + Extra, Aquacel Extra and Convamax, there had been no clinical improvement in the wound’s condition. At the time of assessment, the patient was wearing class II compression hosiery, having been unable to tolerate the standard 40 mmHg hosiery kit. The patient initially presented to community nursing services following a 4-week period of unsupported self-management. Although placed under the care of community nurses, she demonstrated reluctance to adhere to recommended compression therapy. At 6 months, due to the wound’s lack of healing progression, she was referred to Tissue Viability services for further assessment and management.
At initial presentation to the tissue viability service, there were no signs of clinical infection; however, the peri-wound skin appeared unhealthy and fragile. The wound measured 7 cm in length and 6 cm in width, with a wound bed composed of approximately 90% granulation tissue and 10% slough. A high volume of haemoserous exudate was present, accompanied by a slight malodour. Pain was a significant issue, with the patient reporting a score of 9/10, on a scale of 0–10 (0=no pain, 10= extreme pain) and a reduced quality of life (QoL) recorded at 42/70 using the Tickle (2018) unpublished QoL tool [Box 1].
District nursing staff implemented the Isle of Wight Lower Limb Pathway, recommending the UrgoStart Plus Treatment Range [Figure 3]. The UrgoStart Plus Treatment Range is one of the few wound management products to receive a positive Medical Technologies Guidance from NICE (2019), with a formal recommendation for adoption across clinical practice. The NICE committee (2019) concluded that the use of UrgoStart should not be limited to any specific NHS care setting. However, it was acknowledged that decisions regarding its use would typically be made by a multidisciplinary team or under the guidance of a tissue viability specialist.
Central to its mechanism is nano-oligosaccharide factor, a compound shown to inhibit the activity and reduce the number of matrix metalloproteinases, enzymes known to contribute to delayed healing and wound breakdown in complex wounds (Ren et al, 2014). This innovative compound is integrated with the Technology Lipido-Colloid, the foundational component of all Urgo dressings. Technology Lipido-Colloid promotes a moist wound environment and prevents adherence to the wound bed, facilitating pain-free dressing changes. UrgoStart Plus Pad dressings feature polyabsorbent fibres designed to continuously debride and actively cleanse the wound bed, supporting faster and more efficient healing.
A new treatment plan was initiated using UrgoStart Plus Pad as the primary dressing and UrgoKTwo compression therapy. The wound and surrounding skin were thoroughly cleansed and debrided. A superabsorbent secondary dressing was applied, and emollients were used to protect the peri-wound skin.
Week 3: Progress
By week 3 of treatment, there were significant clinical improvements. The wound had reduced in size to 4 cm × 2 cm. Oedema had diminished considerably, and limb shape distortion had resolved. Importantly, there were no signs of wound infection [Figure 4].
Pain levels decreased to a score of 5, and the patient reported reduced stress and anxiety. The QoL score improved to 22, reflecting a positive response to the intervention.
Week 7: Outcome
By week 7 of treatment, the wound had completely healed [Figure 5]. There had been no episodes of infection throughout the treatment period.
Following treatment with UrgoStart Plus and UrgoKTwo, the patient experienced marked symptomatic relief, no longer requiring regular analgesia for pain management. The patient’s pain score reduced to 0, and compression bandaging was transitioned to Altipress hosiery for ongoing care. The intervention led to a sustained improvement in the patient’s QoL, reflecting enhanced physical comfort, mobility and social engagement. The patient resumed normal daily activities and reported a significant improvement in overall well-being, noting improved personal independence and greater ability to participate in family and social roles. This outcome highlights the holistic impact and clinical effectiveness of the care pathway.
Prior to initiating treatment with UrgoKTwo, the patient’s dressing regimen consisted of Aquacel Ag + Extra, Aquacel Extra and Convamax, alongside a long-term compression hosiery kit. This extended treatment approach, spanning up to 24 weeks, incurred a total material cost of £327.15 [Table 1].
In contrast, the structured introduction of UrgoKTwo compression and UrgoStart Plus Pad dressings over a 7-week period resulted in complete wound healing, significantly reduced pain and improved quality of life, with a total spend of £152.46 [Table 2]. The shortened treatment duration demonstrated cost savings of £174.69. These savings did not include reduced reliance on analgesia, reduced practice nursing visits and transition to maintenance hosiery, which reflect clinical efficiency and further cost savings.
Discussion
Wounds remain a major burden to patients and a substantial cost to global health care organisations. The development and implementation of standardised wound care protocols, including guidance documents, pathways and clinical decision-making algorithms, can provide healthcare professionals with a consistent and evidence-based framework for wound assessment and treatment (World Union of Wound Healing Societies, 2020; Smet et al, 2024). Clear evidence-based pathways promote uniformity in clinical practice, improve diagnostic accuracy and facilitate timely interventions. Evidence-based care integrates the best available clinical evidence with professional expertise, patient preferences, and the realities of available resources (Dawes et al, 2005). Tickle (2021) asserts that every individual with a wound is entitled to evidence-based practice where interventions should not only support healing but also promote an improved QoL. Familiarity with less effective treatments should not impede the adoption of superior, more clinically effective alternatives. To ensure optimal patient outcomes, consistency in care delivery is vital.
Implementation of a standardised pathway has exemplified the value of implementing a standardised care pathway supported by advanced wound care technologies. The intervention not only improved patient outcomes but also reduced pain, enhanced her quality-of-life scores and restored daily independence. Notable economic benefits were demonstrated by streamlining resource use and reducing the duration and complexity of treatment.
The implementation of a standardised wound care pathway, supported by advanced technologies, has clearly demonstrated its value in improving both clinical and economic outcomes. Patient benefits included significant pain reduction, enhanced quality of life and promotion of daily independence. These outcomes highlight the pathway’s effectiveness in delivering person-centred care through timely, evidence-based interventions. In addition to clinical improvements, the approach demonstrated notable economic advantages by streamlining dressing selection, optimising resource use, and shortening the duration and complexity of treatment