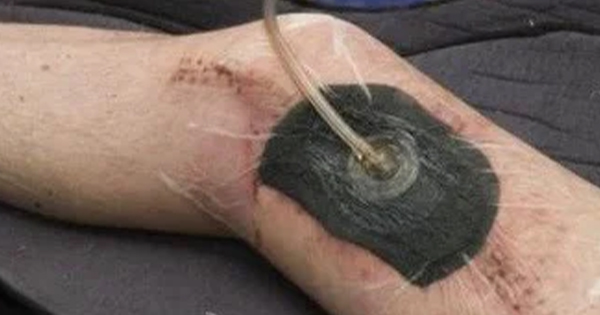
In 2017, the European Wound Management Association (EWMA) highlighted negative pressure wound therapy (NPWT) as a treatment of choice to promote granulation tissue over exposed bone and tendon, where reconstructive procedure is associated with poor functional results (Apleqvist et al, 2017). EWMA updated this NPWT document in 2024 and highlighted that NPWT helps in the stimulation of granulation over bradytrophic structures such as exposed bone and tendon (Apleqvist et al, 2024).
Wounds with exposed bone and tendon are composed of fibrous connective tissue with poor blood supply to the wound bed, thus not providing a suitable environment for flap reconstruction (Deng et al, 2023). Therefore, these types of wounds require healthy granulation tissue at the wound bed for successful flap coverage (Deng et al, 2023). NPWT creates microdeformation in the wound bed through the movement of fluid, fibroblast proliferation and angiogenesis, causing granulation tissue to form over exposed bone and tendon (Quacinella et al, 2022).
This paper primarily focuses on evidence published within the last 7 years to highlight the most recent and clinically relevant literature on NPWT. NPWT has been applied in both adult and paediatric populations. In adults, positive outcomes have been reported with NPWT in open fractures, such as Gustilo Anderson Type 3B, while in children, it has been used in traumatic injuries as an atraumatic therapy with minimal complications. The following section discusses the application of NPWT in open fractures and its effectiveness in promoting granulation over exposed bone and tendon.
Open fractures and NPWT
Open fractures, such as Gustilo Anderson Type 3B, represent significant damage to soft tissue, resulting in wounds with exposed bone and tendon (Miamin et al, 2023). Immediate soft tissue reconstruction with flap coverage is considered standard for management. However, primary coverage of exposed structure is associated with complications, such as wound infection, flap failure, non-union and delayed amputation.
NPWT is regarded as a mainstay for the management of wounds with exposed bone and tendon as it temporises the wound and prepares the wound bed for successful flap coverage (Quacinella et al, 2023). This aligns with EWMA recommendations, which support NPWT as a primary treatment option when flap reconstruction is likely to produce poor outcomes (Apleqvist et al, 2017).
In an observational analytical study by Kumar et al (2022), NPWT was used in 34 patients (mean age 37.06 years) with open Gustilo Anderson 3A/3B limb fracture, of which most were 3B (64.71%). The wounds were classified with the open wound grading system (Lee et al, 2009) as Grade 2 (any bone, tendon, implant exposure) in 52.9% of patients and Grade 3 (combination of bone, tendon or implant exposure) in 35.3% of patients. The authors used NPWT at a pressure of −125mmHg over a clean, debrided wound after fixation of the fracture. In this study, the majority of injuries were due to road traffic accidents, and no neurovascular defects were noted.
Kumar et al (2022) found statistically significant reductions in wound area (p<0.001) woth NPWT. They concluded that NPWT significantly reduced wound size and the need for free flap surgery. Since the study was limited to the experimental group only, it was not possible to analyse the effect of other dressings compared with NPWT.
Hakim et al (2024) conducted a prospective non-randomised control trial to compare NPWT with conventional wet-to-moist saline dressing. The study included patients >18 years old with unilateral Gustilo Anderson 3B tibial fracture where primary coverage of exposed structures was not possible. This study initially assessed all patients treated with Advanced Trauma Life Support protocol followed by radiographic evaluation and fracture stabilisation in a 2-year period. Patients were divided into two groups, where primary coverage of wound with exposed bone and tendon was not possible. The author applied NPWT at a pressure of −125mmHg with a wound dressing change every 4 days in one group (n=20), while the other group (n=20) received regular dressings until healthy granulation was observed in all patients. The authors found that the need for secondary debridement surgeries and time until definitive wound coverage was significantly lower in the NPWT group (p<0.001). The author concluded that patients treated with NPWT successfully underwent coverage procedures after the formation of healthy granulation tissue, whereas four cases in the regular dressings group failed due to a high incidence of infection. However, this study is limited by non-randomisation, which could be a source of bias.
Overall, the studies by Kumar et al (2022) and Hakim et al (2024) highlighted the potential advantage of NPWT for the management of wounds with exposed bone and tendon with statistically significant results. Further randomised control trials are needed to provide more definitive evidence in the use of NPWT for granulation of Gustilo Anderson 3B wounds with exposed bone and tendon. The evidence reviewed by Kumar et al (2022) and Hakim et al (2024) were narrow in focus, addressing only patients >18 years old. In contrast, paediatric wounds are more challenging to manage due to exposed bone and tendon (Slavkovic et al, 2023).
Wounds with exposed bone and tendon in children
Reconstructive procedures for wounds with exposed bone and tendon are more challenging in children due to the risk of anastomosis and thrombosis (Rasool et al, 2020). However, the atraumatic nature of NPWT and fewer dressing changes make it a valuable option for managing wounds with exposed bone and tendon.
Rasool et al (2020) conducted a prospective study design where NPWT was used in 46 children (mean age 8.4 years) treated between July 2016 to June 2018 in a plastic surgery department in India. The authors included patients with injuries from road traffic accidents (48%), machines (24%) and falls from height (11%). Patient wounds with exposed bone (n=24) and exposed tendon (n=15) were treated with NPWT at a pressure of −125mmHg until healthy granulation was seen over exposed structures. Healthy granulation with 90% coverage of developed in 89% of cases, while only four cases had <50% coverage. Minor complications were seen, including pain, peri-wound maceration and bleeding. However, none of the complications led to the discontinuation of NPWT therapy. Although the study is limited to the experimental group, the findings suggest NPWT as an atraumatic therapy for granulation of wounds with exposed bone and tendon in children with significant results.
Klein et al (2021) conducted a retrospective continuous single-centre cohort study for children, treated from 2001 to 2016, in a paediatric orthopedic surgery department in France. The authors included 18 children (mean age 4 years 10 months) with severe foot and ankle injuries associated with soft tissue defects. In the department before 2003, wounds with exposed bone and tendon were covered with latissimus dorsi free flap or local flap. However, after 2003, the procedure was modified and exposed structures were treated with NPWT to encourage granulation tissue. This study used continuous NPWT of −125mmHg. The author found healthy granulation tissue in all cases followed by successful reconstructive procedure as artificial dermis in six patients and skin grafts in seven patients. Klein et al (2021) concluded that NPWT had improved the management of these injuries in children with minor complications.
Although both studies by Rasool et al (2020) and Klein et al (2021) were limited to their experimental groups, their findings are less generalisable for analysing the effect of NPWT on other conventional dressings.
Slavkovic et al (2023) conducted a prospective single institute study using 58 paediatric patients to compare NPWT with conventional wet-to-moist saline dressing for wounds with exposed bone and tendon. The author divided patients into two groups with the study group treated with NPWT (n=34) and the control group (n=24) treated with conventional wet-to-moist dressing. Both groups were divided into two subgroups: traumatic wounds and surgical wounds. In this study, most traumatic wounds were due to road traffic accidents, followed by agriculture machinery wounds in the NPWT group and sharp surgical object wounds in the control group. However, most surgical excision wounds were due to soft tissue infections. NPWT was used at a continuous negative pressure of -100 mmHg to -125 mmHg with dressing changes at 3-5 day intervals under general anaesthesia until healthy granulation was seen over exposed structures. The author found that patients in the NPWT group were older than the control group, while wounds treated with NPWT were slightly larger than the control group with exposed bone in 17 patients and tendon in 9 patients. Patients treated with NPWT stayed longer in the hospital (p=0.002) due to taking more time from injury to the start of treatment, but with fewer dressing changes (p=0.009). Minor complications were seen in eight patients in the NPWT group after discontinuation of therapy as compared to a control group with no minor complications, while two major complications were seen in both cases. The authors concluded that NPWT is effective in terms of fewer dressing changes and makes it possible for definitive wound coverage with simpler technique while wet to moist saline dressing is effective in terms of shorter hospital stay. They emphasised the significance of the study findings, as it is the first prospective study to compare NPWT with conventional wet-to-moist saline dressings in a substantial patient population.
Electrical injury wounds and NPWT
Electrical injury wounds with exposed bone and tendon have also shown promising results, with healthy granulation observed in wounds caused by high-voltage electric currents.
Evyaz et al (2018) conducted a retrospective study using NPWT in electric burn wounds with exposed bone and tendon. The author used a prospective database of electric burn patients treated in a burn centre between August 2008 and December 2012. The study included electric burn patients (mean age 34.9 ± 9.8 years) where primary grafting was not possible in 27 cases. Of the 27 cases, 24 achieved healthy granulation over exposed structures with a success rate of 89%. The authors highlighted this study as the first published evidence of NPWT use for electrical burn wounds with exposed bone and tendon (Evyaz et al, 2018).
Discussion
Overall, the recent evidence highlighted represents the significance of NPWT for the granulation of wounds with exposed bone and tendon, with statistically significant results. However, large, randomised control trials are needed to determine the generalisability of these results.
Despite the clinical benefits of NPWT, the high cost associated with the device comes up with a significant barrier to its use in low- and middle-income countries (LMICs, Farré et al, 2022). In these countries, where healthcare financing is out of pocket, the feasibility and affordability of NPWT devices are challenging. A fundamental change is required to implement NPWT devices through a dedicated funding mechanism and the introduction of low-cost NPWT devices in LMICs(Farré et al, 2022).
The financial burden associated with NPWT limits its use, particularly in public clinical settings with constrained budgets, especially for underinsured or uninsured patients (Kim et al, 2017). While in long run, it provides cost-effectiveness in terms of less dressing changes, reducing nursing time and material cost, the upfront expenses out weights long-term savings, limits its use in hospitals. This emphasises the need for government-funded programmes and low-cost NPWT innovations to enhance clinical affordability, particularly in low- and middle-income countries.
Continued efforts are needed to make NPWT more accessible to patients through dedicated funding mechanisms in LMICs. In Pakistan, there are several government-funded mechanisms for medical treatments but the use of NPWT is limited under these programs (Khan et al, 2023). Through advocacy and policy, the use of NPWT should be implemented in these programs highlighting the cost effectiveness of NPWT in terms of lesser dressing changes and shorter hospital stay as compared to conventional dressings. Moreover, the implementation of low-cost NPWT could further enhance affordability for patients.
Conclusion
NPWT is highlighted as a mainstay for the granulation of wounds with exposed bone and tendons where primary coverage or repeated dressing changes are not possible. However, at present NPWT devices should be made easily accessible and affordable to patients through advocacy and policy. Moreover, there is a need for a large, randomised controlled trial to provide more robust evidence; therefore, further research needs to be carried out.